The DNA in the billions of cells within the human body is under constant assault from various threats, both internal and external, collectively known as genotoxic stress, which can lead to DNA damage. If left unrepaired, this damage can result in genome instability, a significant factor in the development of cancer and various genetic diseases.
Led by Assistant Professor Ching-Shyi Peter Wu from the NTU Department and Graduate Institute of Pharmacology, a research team has made a groundbreaking discovery. For the first time, they have demonstrated that ATR, a pivotal protein kinase in the DNA damage response, plays a crucial role in preventing genotoxic stress-induced genome instability by regulating the RNA helicase DHX9, a critical RNA processing factor with functions in several physiological processes, including transcriptional regulation, RNA splicing, and R-loop resolution.
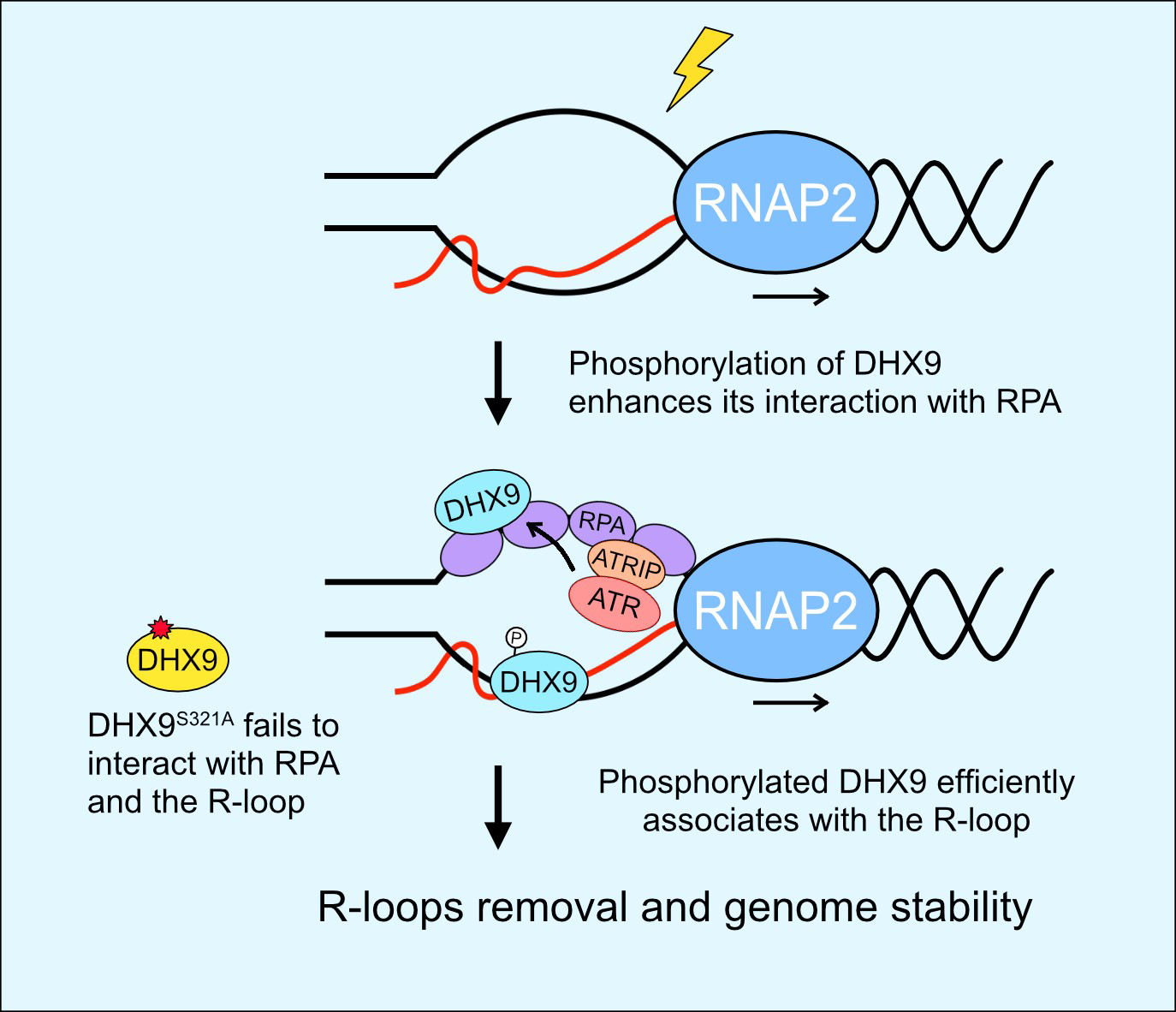
ATR acts as a key kinase in the DNA damage response, becoming activated when cells encounter genotoxic stress. Once activated, ATR phosphorylates proteins involved in the DNA damage response, allowing them to maintain genome stability. Notably, during DNA replication and transcription, genotoxic stress can trigger the formation of R-loops, contributing to DNA damage and genome instability.
DHX9, an RNA helicase, has been identified as a key factor in reducing R-loop accumulation, thereby preserving genome stability. In their recent study, the research team discovered that ATR directly phosphorylates DHX9 to reduce the accumulation of R-loops during genotoxic stress. Phosphorylation of DHX9 at S321 enhances its interaction with DNA damage proteins γH2AX, BRCA1, and RPA32, facilitating its association with R-loops in response to DNA damage. More importantly, compromised phosphorylation of DHX9 at S321 leads to increased R-loop accumulation and hypersensitivity to genotoxic drugs in cells. These findings establish a critical connection between ATR, RPA, and DHX9 in responding to DNA damage-induced R-loops, highlighting ATR's direct regulation of RNA helicase DHX9 to prevent genome instability. Insights into the molecular mechanisms governing DHX9's function and its dysregulation in diseases offer promise for the development of targeted therapeutic interventions and diagnostic approaches.